Pharmacologic THC and THC derivatives
In the USA, the FDA has approved several cannabinoids for use as medical therapies: dronabinol (Marinol) and nabilone. These medicines are taken orally.
These medications are usually used when first line treatments for nausea and vomiting associated with cancer chemotherapy fail to work. In extremely high doses and in rare cases “psychotomimetic” side effects are possible. The other commonly used antiemetic drugs are not associated with these side effects.
Canasol is a cannabis-based medication for glaucoma that relieves intraocular pressure symptoms associated with late-stage glaucoma.
It was created by an ophthalmologist, Dr. Albert Lockhart and Dr. Manley E. West, and began distribution in 1987.[97][98] As of 2003, it was still being distributed in the United Kingdom, several US states, and several Caribbean nations.[99]
It is notable for being one of the first cannabis-containing pharmaceuticals to be developed for the modern pharmaceutical market and being one of the few such pharmaceuticals to have ever been legally marketed in the United States.[98][100]
The prescription drug Sativex, an extract of cannabis administered as a sublingual spray, has been approved in Canada for the adjunctive treatment (use along side other medicines) of both multiple sclerosisand cancer related pain.[101][102] Sativex has also been approved in the United Kingdom, New Zealand, and the Czech Republic, and is expected to gain approval in other European countries.[103][104][105]William Notcutt is one of the chief researchers that has developed Sativex, and he has been working with GW and founder Geoffrey Guy since the company’s inception in 1998. Notcutt states that the use of MS as the disease to study “had everything to do with politics.”[106]
| Medication | Approval | Country | Licensed indications | Cost |
|---|---|---|---|---|
| Nabilone | 1985 | USA, Canada | Nausea of cancer chemotherapy that has failed to respond adequately to other antiemetics | US$ 4000.00 for a year’s supply (in Canada)[107] |
| Canasol | 1987 | USA, Canada, several Caribbean nations | Introcular pressure associated with late-stage Glaucoma | |
| Marinol | 1985 | USA Canada (1992) |
Nausea and vomiting associated with cancer chemotherapy in patients who have failed to respond adequately to conventional treatments | US$ 652 for 30 doses @ 10 mg online[108] |
| 1992 | USA | Anorexia associated with AIDS–related weight loss[109] | ||
| Sativex | 1995 | Canada | Adjunctive treatment for the symptomatic relief of neuropathic pain in multiple sclerosis in adults | C$ 9,351 per year[110] |
| 1997 | Canada | Pain due to cancer |
Difference between Cannabis indica and Cannabis sativa
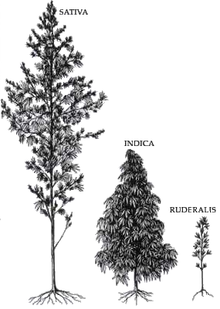
Cannabis sativa, Cannabis indica, andCannabis ruderalis
A Cannabis indica plant may have a CBD/THC ratio 4–5 times that of Cannabis sativa. Marijuana and Medicine: Assessing the Science Base (1999) states that “There are numerous anecdotal reports claiming that marijuana with relatively higher ratios of THC:CBD is less likely to induce anxiety in the user than marijuana with low THC:CBD ratios; but, taken together, the results published thus far are inconclusive.” (Page 36)[111] Indica has more cannabidiol and sativa has more THC.[112]This might partially be due to CBD’s antagonist effects at the cannabinoid receptor, compared to THC’s partial agonist effect.[113] The relatively large amount of CBD contained in Cannabis indica, means, compared to an Cannabis sativa, the effects are modulated significantly. The effects of sativa are well known for its cerebral high, hence used daytime as medical cannabis, while indica is well known for its sedative effects and preferred night time as medical cannabis. Indica plants are normally shorter and stockier plants than sativas. They have wide, deeply serrated leaves and a compact and dense flower cluster. The effects of indicas are predominantly physical and sedative. Due to the relaxing nature of indicas, they are best used for non-active times of the day, and before bed. Indica strains generally have higher levels of C.B.D and C.B.N and lower levels of T.H.C.[114]


